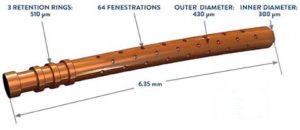
The CyPass Micro-Stent is a small medical device used at the time of cataract surgery to lower pressures in patients with glaucoma.
Glaucoma is a medical condition where the pressure inside the eye is too high, leading to progressive optic nerve damage. Open angle glaucoma, the most common form of glaucoma, is essentially a backing-up of fluid due to insufficient outflow through the natural drainage system.
In the eye, there are two distinct drainage systems: the traditional trabecular meshwork, and the uveoscleral pathway. Many of the more potent glaucoma eye drops work to decrease eye pressure by increasing the flow through the uveoscleral pathway.
The CyPass stent works in this same method. It is inserted into the uveoscleral pathway, creating a conduit for the fluid within the eye to exit with less resistance. There are three retention rings (see above diagram), which help to guide placement and provide stability for the buried CyPass stent. Fenestrations at the tip of the stent allow fluid to drain, effectively lowering the intraocular eye pressure.
In the US, the CyPass Micro-Stent is FDA approved to be used to treat mild to moderate glaucoma in patient who are undergoing simultaneous cataract surgery. It is considered a Minimally Invasive Glaucoma Surgery (MIGS) device, which means it may not be as effective as some of the more traditional aggressive glaucoma surgeries, but it carries significantly less risk.
However, in August of 2018, the CyPass Micro-Stent was voluntarily removed the market due to long-term safety concerns. In a select few patients where the Micro-Stent was not inserted deeply enough, the protruding stent seemed to cause degradation of the adjacent corneal tissue. It is recommended that patients who have had the CyPass Micro-Stent inserted be monitored on a regular basis by their ophthalmologist.